
Features
Agronomy
Soil
Assessing Group 2 herbicide carryover
Group 2 herbicides, like the ALS inhibitors in the imidazolinone, sulfonylurea, triazolopyrimidine sulfonanalide, pytimidinylthiobenzoate, and sulfonylaminocarbonyl-triazolinone categories, have the potential to carryover in the soil and affect rotational crops.
December 3, 2008 By Bruce Barker
A new diagnostic tool can help with rotational decisions.

|
|
| Group 2 carryover can inhibit root and shoot growth.Photos courtesy of University of Saskatchewan.
|
Group 2 herbicides, like the ALS inhibitors in the imidazolinone, sulfonylurea, triazolopyrimidine sulfonanalide, pytimidinylthiobenzoate, and sulfonylaminocarbonyl-triazolinone categories, have the potential to carryover in the soil and affect rotational crops. The extent to which re-cropping injury may occur from Group 2 herbicide carryover depends on a host of factors including the herbicide, its rate of application, sensitivity of crop grown in rotation, soil and environmental conditions. “Carryover of a herbicide beyond the year of application can be of benefit in controlling weed growth in subsequent years, but can also be of concern in causing injury to sensitive crop species that may be grown in the years following application,” says Jeff Schoenau, a soil scientist at the University of Saskatchewan.
Group 2 activity is often measured in the parts per billion or less range. Damage often appears as root stunting and pruning due to the growing point being affected, and can ultimately result in reduced biomass or economic yield.
Schoenau explains that the phytotoxicity of a soil residual herbicide refers to the toxic effect that the residue has on plant growth. Phytotoxicity is dependent on the amount of herbicide that sorbs to the soil particles and the rate at which it can desorb back into the soil water.

|
|
| Eroded knolls often have greater phytotoxicity and persistence of residues. |
Factors to consider
Organic matter and clay content are the two components of soil largely responsible for herbicide sorption. Many studies have shown that soils with high organic matter and clay content result in increased sorption and reduced phytotoxicity of Group 2 herbicide compounds like imazethapyr, flucarbazone and many sulfonylurea herbicides, says Schoenau. While the sorption of a herbicide will reduce its phytotoxicity by removing it from the soil solution, it can also increase its persistence or half-life (time for 50 percent of the herbicide to disappear), as the adsorption can render the compound unavailable for degradation. Different herbicide compounds have different rates of degradation in the soil, with reported half – lives ranging from two days to over 100 days, depending on the compound.
Soil properties such as organic matter content, soil texture and soil pH play a role in the carryover of residual herbicides. “From our research, organic matter content seems to be an important factor in prairie soils. High organic matter usually means lower risk of herbicide injury,” explains Schoenau. Higher pH can result in reduced sorption of some Group 2 herbicides like sulfonylureas and produce greater phytotoxicity and injury. On the other hand, Schoenau says that high pH and reduced sorption can sometimes result in greater degradation rates and reduced persistence such as for the imidazolinone herbicides.
Soil temperature and moisture also have a very important effect on the rate of microbial decomposition. In cooler and drier soils, microbial degradation rates are reduced. As a consequence the risk of carryover and injury in recropping is increased with lower than average growing season precipitation and temperature. That’s the reason that some Group 2 herbicides have recropping restrictions outlined on their label that are sensitive to growing season precipitation and temperature in the year of application, along with soil properties like organic matter and pH.
Since herbicide carryover and injury is sensitive to such a wide range of variables, Schoenau says that it is not surprising that recropping injury tends to be variable across farm fields, owing to variations in soil organic matter content, texture, pH, moisture and temperature.
Citing a farm example, Schoenau says that injury tends to be greatest in the upslope areas of fields, especially on eroded knolls where a combination of low organic matter content, high pH, and lower soil moisture content combine to result in greater phytotoxicity and persistence of residues. “Generally, if soils are warm and moist with high organic matter and high clay content, those soils will have the least carryover,” he explains.
 |
|
|
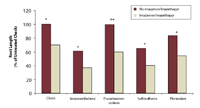
Oriental mustard root length responses as a percent of the untreated check averaged over six site years in three Saskatchewan soils from samples taken one year after the application of four herbicides (if applied) and two years after the application of imazamox/imazethapyr (if applied). Source: B. Geisel, MSc thesis, University of Saskatchewan
|
Stacking can result in additive carryover
When a residual herbicide is applied in one year, and then another residual herbicide applied in the subsequent year, the possibility of ‘stacking’ of these herbicides to cause carryover injury occurs.
Laboratory and field work conducted at three sites in Saskatchewan on soils collected from treatments with application of the herbicide imazamox/imazethapyr to peas, followed by either imazamethabenz, flucarbazone, sulfosulfuron, or florasulam to wheat the next year revealed no antagonistic (decreased) or synergistic (more toxicity) interactions of the herbicide residues.
However, injury from sequential field applications of ALS inhibiting herbicides over two years was additive (sum of the individual affects), as assessed by root length inhibition in a mustard root length bioassay.
Schoenau cautions that this research implies that the potential is still there for greater phytotoxic effects when two separate residual herbicides are applied as compared to one. “There is the possibility for rotational crops to be injured from two residues present and acting together in an additive manner compared to only one residue present if the rotational crop is sensitive to both compounds. It may be desirable to avoid sequential application of residual ALS inhibiting herbicides over two years if sensitive crop species are grown in rotation,” explains Schoenau.
New bioassay test assessed
A simple mustard root length bioassay was developed at the University of Saskatchewan for detection of ALS inhibitor residues in soil. The bioassay, developed by Dr. Szmilgielski, measures the inhibition of shoot growth and root length of mustard plants grown in bags of soil. This bioassay is completed in three days using 200 g of soil for four replicate measurements.
Schoenau says the bioassay was field tested to assess the accuracy. They found good agreement between yield reductions in treated field soils and the mustard root length bioassay. However, there were approximately five to 12 percent of the samples that showed a false negative result – meaning that the bioassay did not show herbicide carryover while the field test showed yield reductions. “The bioassay has some application as a warning flag for an agronomist. It can help show that something is there that the farmer needs to be concerned about, but it isn’t the final word on whether residues are there at levels that will affect the rotational crop,” says Schoenau. “It is a good diagnostic tool, but many environmental and soil factors can affect the extent to which injury occurs in the field and cannot be measured or reliably predicted in a bioassay conducted on a sample of soil.”
When considering the potential for carry over, Schoenau summarizes by saying that farmers and agronomists should watch out for dry conditions, especially on sandy soils with low organic matter. Stacked herbicides may also be a concern. He says that detection and prediction bioassay tools are getting better, but that label directions on recropping intervals for sensitive species should be followed. n