
Features
Agronomy
Soil
Fall application of nitrogen fertilizer
The effectiveness of fall fertilizer applications depends on a number of factors, including the form of nitrogen (N) fertilizer used, how the fertilizer is applied and, most important, the environmental conditions after application including soil moisture and temperature.
Fertilizer nitrogen basics
The four most common nitrogen fertilizers used in Western Canada are urea 46-0-0 [CO(NH2)2], anhydrous ammonia 82-0-0 [NH3], liquid N 28-0-0 which contains half urea and half ammonium nitrate [ammonium (NH4+) and nitrate (NO3-)], and ESN 45-0-0 [Environmentally Smart Nitrogen – slow release urea with a polymer coated granules]. When urea and anhydrous ammonia are banded into warm, moist soil, both convert to ammonium [NH4+]. Ammonium is positively charged and is relatively immobile in soil and will not leach under wet conditions. In warm, moist soil, specific bacteria will convert ammonium to nitrate over a two- to three-week period. This process is called nitrification. Nitrate is negatively charged, is mobile in soil and will leach with excess precipitation, particularly in sandy soils.
Banding ammonia or urea creates an environment near the band that slows the activity of bacteria that convert ammonium to nitrate, delaying nitrification. When urea or anhydrous ammonia are banded in late fall after the soils have cooled and micro-organism activity has slowed, most of the fertilizer N remains in the ammonium form over winter until the soil warms up in the spring. Over the winter and in the early spring, when soils are cold, the ammonium form is relatively stable and won’t leach.
However, if urea or anhydrous ammonia fertilizers are banded in early fall when soils are still warm and moist, much of the ammonium can potentially be converted to nitrate before freeze-up. Excess precipitation in late fall or spring could then cause the nitrate to leach below the crop root zone or be lost due to a process called denitrification. The denitrification process occurs after N fertilizer has converted to nitrate, the soils are warm and soil conditions become saturated after snow melt in spring or due to heavy precipitation events and soils. Soil nitrate-N is lost when microorganisms in anaerobic conditions (saturated soil without oxygen) convert nitrate N to nitrous oxides which is a gaseous form of N that is lost to the atmosphere.
Research has shown that denitrification can occur in virtually all western-Canadian agricultural soils. Denitrification is caused by many different types of soil bacteria that use the process to obtain oxygen from nitrate, when oxygen is in short supply in very wet soils.
All soil types and regions of the prairies are susceptible to losses of fall-applied N fertilizer. However, the risk of N loss is typically highest in regions with moister climates, causing soils to be more frequently saturated with water, such as the Black and Gray soil zones. Denitrification is usually lowest in regions that tend to be drier, such as the Brown and Dark Brown soil zones.
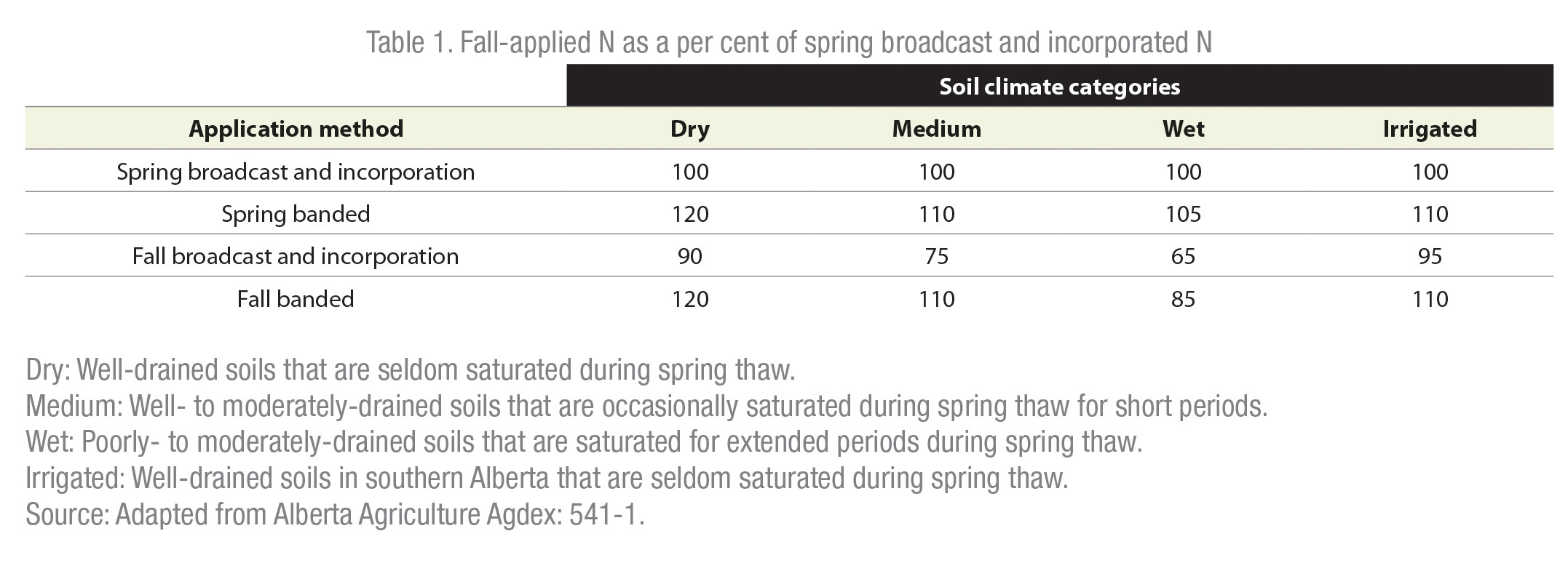
Nitrogen losses in drier regions are usually low, and, therefore, fall-banded N is usually as equally effective as spring-banded N (see Table 1). However, N losses can still be high even in low-risk regions, after heavy precipitation events. Each fall, farmers must look at their specific environment conditions on their farms to weigh the risks versus benefits of fall fertilizer application. In cases where spring banding causes a significant loss of seedbed moisture, fall banded N can produce a higher yield than spring banded N.
Nitrogen fertilizer applied in the fall can be very effective on well-drained land, but can be totally ineffective in poorly drained soils and low-relief soils that tend to be wet for extending periods during the spring. Even during drier springs, localized lower areas in fields that are wet are subject to denitrification. It is important to remember that fall application always puts your fertilizer N at risk. Assess the level of risk first at the regional level based on typical long-term environment conditions and then based on specific, very local field conditions each fall.
Some general rules of thumb to consider:
- Spring banding can be the more effective method of application, and fall broadcast the least effective when soils are wet for longer periods in the spring or if land is moderately rolling with numerous low areas that stay wet for prolonged periods.
- Late fall-banded N can be as effective as spring-banded N, if there is no extended period of saturated soil conditions in the spring.
- Fall-banded N can be more effective than spring-banded when springtime seedbed moisture is limited, and spring banding would dry out the seedbed.
Management recommendations
General tips to consider before fall banding N:
- When soils are poorly drained and tend to be saturated with water for extended periods in the spring, then fall application is generally not a good option. However, if saturated soil conditions are normally not a problem, fall banding can be an effective N application method.
- Generally under lower-risk conditions, such as in the Brown and Dark Brown soil zones, anhydrous ammonia (82-0-0) and urea (46-0-0) perform equally well when properly fall banded. However, if soils tend to be alkaline (soil pH >7.5), losses through ammonia volatilization can occur if the bands are too shallow or the soil is dry and cloddy.
- Apply a conservative N fertilizer rate of about 75 per cent of the crop requirement in the fall. This reduced fall rate is a hedge against potential over-winter losses, low spring moisture or lower crop prices in the following spring. If conditions look favourable in the spring, additional N can be applied at seeding.
- Avoid the use of the nitrate-containing fertilizer products for fall application such as 28-0-0, as the nitrate is subject to both denitrification and leaching losses under wet conditions in late fall or spring conditions.
- Apply N as late as possible in fall after the soil temperature has dropped below 5 to 7 C and the nitrification process has slowed down.
- It is best to band and not broadcast/incorporate N fertilizer. Banding restricts the contact between soil and fertilizer, concentrating the band delays nitrification. Consequently, over-winter N losses are lower with banded than with broadcast/incorporate N.
Advantages of fall nitrogen application
Fall fertilization shifts the workload from the hectic spring to the fall. This can increase spring seeding operation efficiency by applying the majority of N fertilizer in fall, and fertilizer handling in spring is reduced at seeding.
Nitrogen fertilizer prices tend to be lower in the fall than in the spring, providing an economic advantage with fall versus spring fertilization. Generally, soils also tend to be drier in the fall than in the spring, reducing potential for soil compaction problems.
It is always wise to get several opinions from experts in your region, including your fertilizer dealer, industry agronomist and government agronomist, to consider all the pros and cons of fertilizing in the fall before you make a final decision
 |
September 3, 2014 By Ross H. McKenzie PhD P. Ag.